Clinical Studies without Discovery
screened
recruited
analysed






Clinical Studies with Discovery
Discovery transforms data workflows, enabling parallel workflows and centralizing data analysis to accelerate clinical study timelines



















SPONSOR: Novartis
STUDY OBJECTIVES:
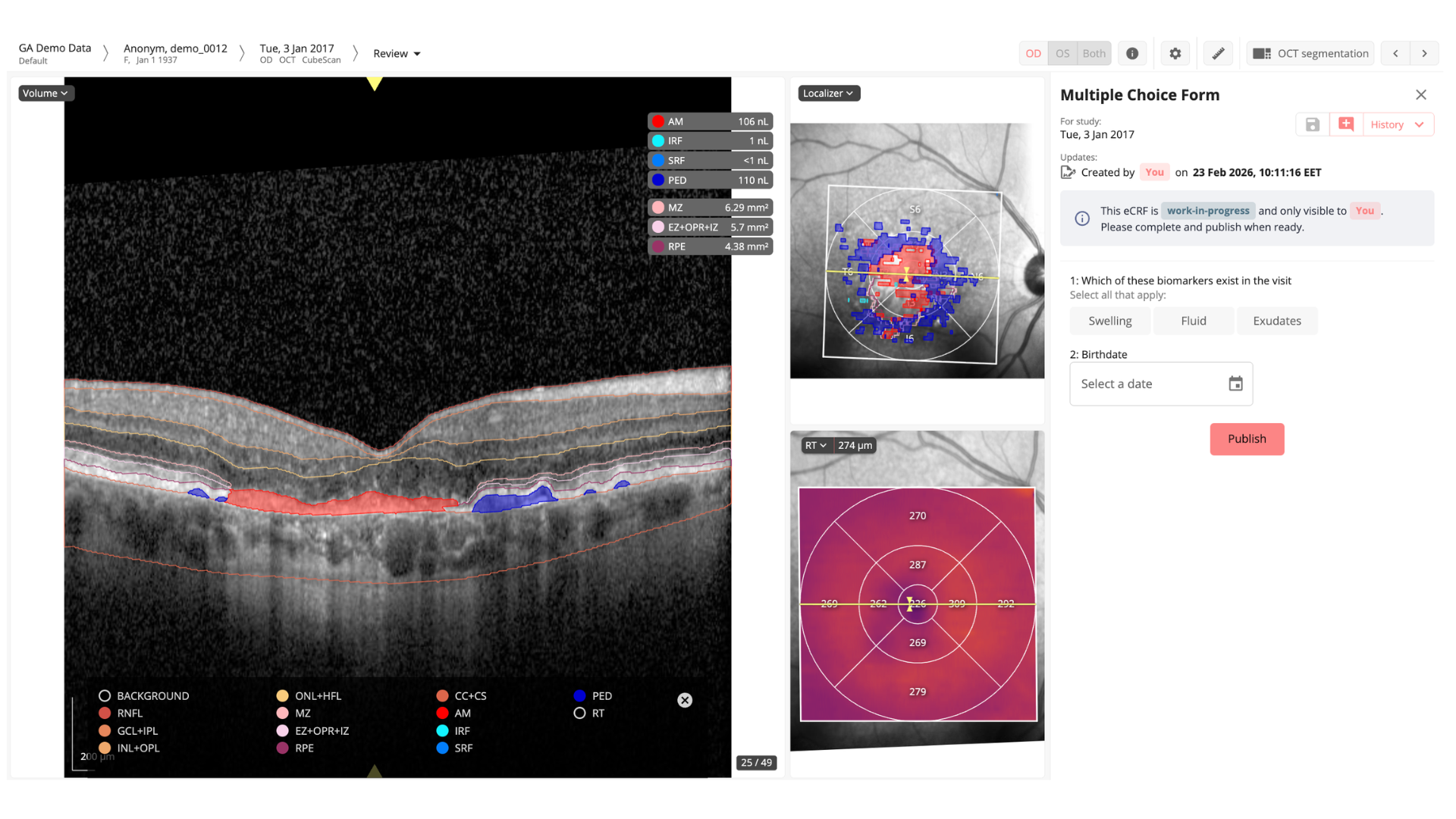
1. Evaluate the influence of automated OCT image enrichment (using AI) on disease activity assessment in nAMD patients treated with anti-VEGF injections
2. Assess if RetinAI’s Discovery can optimize clinical workflows
DISCOVERY’S ROLE:
in phase I of the study → data collection
in phase II of the study → automatic OCT enrichment using RetinAI’s fluid and layer segmentation certified models
SPONSOR: Isarna Therapeutics GmbH
STUDY OBJECTIVE: Evaluate the influence of automated OCT image enrichment (using AI) on disease activity assessment in nAMD patients treated with anti-VEGF injections
DISCOVERY’S ROLE: Provide a same day data evaluation workflow for enrollment by review of the clinical and imaging data between sites and reading center. RetinAI’s fluid and layer segmentation models are used to evaluate the primary endpoints (reduction of fluid and center macula thickness).
STUDY OBJECTIVE: Evaluate the statistical significance (sensitivity and specificity) of the LuxIA algorithm for screening Diabetic Retinopathy, compared to three, independent and blinded graders.
DISCOVERY’S ROLE: Platform was used for the data collection and data review phases. In the data collection phase, Discovery’s eCRFs were used to collect clinical patient data as well as multi-modality imaging data such as fundus images (used for analysis of the algorithm) and OCTs. During the review phase, the workflow deployed in Discovery, ensured graders were blinded to each other’s grading outcomes for robust, unbiased analysis.
*Disclaimer
RetinAI Discovery® platform, RetinAI OCT Atlas® and Macula Biomarkers, as well as LuxIA® (Diabetic Retinopathy Screening model) are CE-marked medical devices in accordance with the Medical Devices Regulation (EU) 2017/745 (CE2797). RetinAI Discovery® platform is also a medical device cleared for clinical use by the FDA. The AI modules for biomarkers, fluid, and layer segmentation and quantification in retinal pathologies are designated as Research Use Only (RUO) in the USA. Please be advised that these tools are not intended to replace professional medical advice, diagnosis, or treatment. We do not warrant or guarantee the clinical accuracy, completeness, or usefulness of any content provided by RUO products. For more information, please read our Privacy Notice (https://www.retinai.com/privacy-policy) and Data Processing Agreement (https://www.retinai.com/data-processing-agreement).
RetinAI Discovery® and RetinAI® are trademarks of Ikerian AG.